Learn About PFAS
Throughout New England and across the nation, state and local officials, health departments, and water utilities, including Aquarion, have focused their attention on a group of man-made chemicals called per- and polyfluoroalkyl substances (PFAS) that have been detected in drinking water. PFAS are widely used in consumer products (e.g. nonstick cookware, stain-resistant carpets) and have numerous industrial applications (e.g. firefighting foam). PFAS are pervasive and persistent once released into the environment.
On March 15, 2023 the US EPA proposed a National Primary Drinking Water Regulation (NPDWR) for 6 PFAS Compounds. Aquarion is continuing our voluntary PFAS testing program in each of our water systems to ensure our customers receive the highest quality water achievable. This proposed rule does not require action until it is finalized in 2024, however, Aquarion will proactively be working to meet the proposed NPDWR.
Explore below to learn more about PFAS.


PFAS reports and FAQs for Aquarion's Connecticut water systems.
Learn more
PFAS reports and FAQs for Aquarion's Massachusetts water systems.
Learn more
PFAS reports and FAQs for Aquarion's New Hampshire water systems.
Learn moreFrequently Asked Questions (FAQs)
The technical name for these compounds are Per- and Poly-Fluoroalkyl Substances (PFAS). Formerly described as perfluorochemicals (PFCs), PFAS is a better scientific and regulatory acronym because it refers only to the compounds found in water.
PFAS are chains of carbon atoms with fluorine atoms attached at most (poly-fluoro-alkyl-substances) or all (per-fluoro-alkyl-substances) of available carbon bonds. The figure below shows per-fluoro-octanoic-acid (PFOS) one of the more common PFAS.
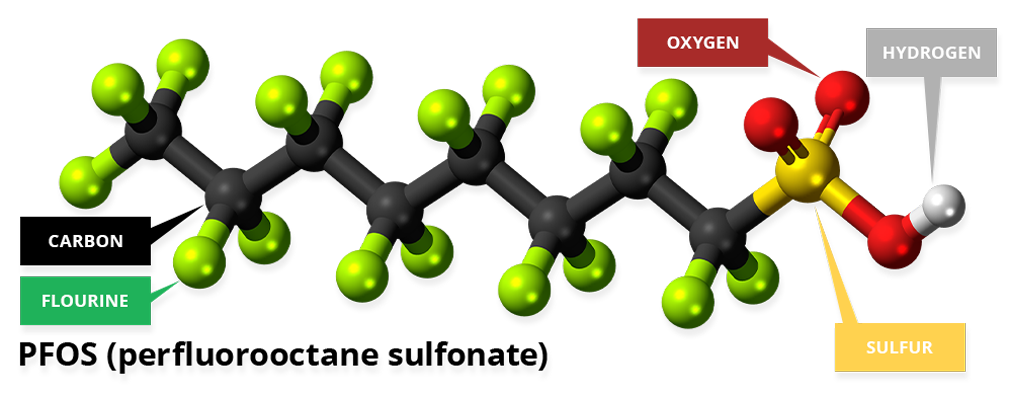
PFAS are a large group of manufactured compounds that are widely used to make everyday products that repel water and oil and are resistant to heat and chemical reactions. Teflon, Stainmaster, and Gore Tex are some of the common trade names for PFAS. PFAS are used in waterproof and stain proof fabrics, non-stick cookware, food packaging, automotive waxes, fire-fighting foams, and in a variety of other industries, including aerospace, automotive, building and construction, and electronics.
Two specific compounds, perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA), are released into the environment when products containing Teflon and other PFAS-bearing materials are made, used, or discarded. PFOS is no longer manufactured in the United States, and PFOA production has been reduced and will soon be eliminated; however, foreign-made products may still contain these compounds.
PFAS have been found in water, air, soil, house dust, and blood. People are exposed to PFAS through food, consumer products, and indoor and outdoor air.
Treatment technologies exist that can remove PFAS from water using granular activated carbon, ion exchange, and other methods.
Aquarion is continuing its PFAS testing program and conducting studies to ensure compliance with all regulatory requirements. When warranted, PFAS removal treatment systems are being implemented. These industrial-scale facilities to remove PFAS are complex and require several years of design and construction before they are operational.
More Information
- Connecticut Department of Public Health (CT DPH) PFAS
- Massachusetts Department of Environmental Protection (MASSDEP) Per- and Polyfluoroalkyl Substances (PFAS) in Drinking Water: Questions and Answers for Consumers
- New Hampshire Department of Environmental Services (NH DES) PFAS Investigation
- Centers for Disease Control and Prevention (CDC) PFAS and Your Health
- Environmental Protection Agency (EPA) Basic Information on PFAS
- Environmental Protection Agency (EPA) Drinking Water (DW) Health Advisories
- American Water Works Association (AWWA) Briefing on PFAS
